Topic 5 Electricity and chemistry
Topic content:
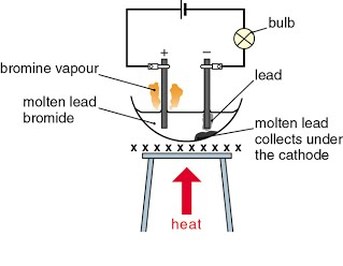
Describe the products of electrolysis of lead(II) bromide, concentrated hydrochloric acid, concentrated aqueous sodium chloride between inert electrodes such as platinum or carbon
The general principle that metals or hydrogen are formed at the negative electrode (cathode) and that non-metals (other than hydrogen) are formed at the positive electrode (anode).
The products of the electrolysis of a binary compound in the molten state
Relate the products of electrolysis to the electrolyte and electrodes used. This should include aqueous copper sulfate using carbon electrodes and using copper electrodes.
Describe the products of electrolysis of lead(II) bromide, concentrated hydrochloric acid, concentrated aqueous sodium chloride between inert electrodes such as platinum or carbon
The general principle that metals or hydrogen are formed at the negative electrode (cathode) and that non-metals (other than hydrogen) are formed at the positive electrode (anode).
The products of the electrolysis of a binary compound in the molten state
Relate the products of electrolysis to the electrolyte and electrodes used. This should include aqueous copper sulfate using carbon electrodes and using copper electrodes.
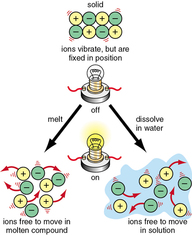
Describe electrolysis in terms of the ions present and the reactions at the electrodes.
Predict the products of electrolysis of a specified halide in dilute or concentrated aqueous solution.
Describe the electroplating of metals.
Name the uses of electroplating
Describe the reasons for the use of copper and (steel cored) aluminium in cable, and why plastics and ceramics are used as insulators.
Describe the manufacture of aluminium from aluminium oxide in molten cryolite and chlorine and sodium hydroxide from aqueous sodium chloride
Predict the products of electrolysis of a specified halide in dilute or concentrated aqueous solution.
Describe the electroplating of metals.
Name the uses of electroplating
Describe the reasons for the use of copper and (steel cored) aluminium in cable, and why plastics and ceramics are used as insulators.
Describe the manufacture of aluminium from aluminium oxide in molten cryolite and chlorine and sodium hydroxide from aqueous sodium chloride
|
|
|
|
|
|
|
Topic 5 Electricity and chemistry Keywords
| topic5electricityandchemistry.docx | |
| File Size: | 6 kb |
| File Type: | docx |
Learning resources:
Some animations on the electrolysis of brine (salt solution)
http://www.absorblearning.com/media/attachment.action?quick=ug&att=218
http://www.absorblearning.com/media/item.action?quick=uf
http://www.absorblearning.com/media/item.action?quick=uh
Movie
http://www.youtube.com/watch?v=w-XbxSiDQWY&feature=PlayList&p=C470A9E10C6186FC&playnext=1&playnext_from=PL&index=45
Another clip about electrolysis
http://www.youtube.com/watch?v=i9xS9t-KMpc&feature=PlayList&p=AA571057652E55EB&index=3
Aluminium extraction simulation
http://www.absorblearning.com/media/attachment.action?quick=u8&att=2166
Some animations on the electrolysis of brine (salt solution)
http://www.absorblearning.com/media/attachment.action?quick=ug&att=218
http://www.absorblearning.com/media/item.action?quick=uf
http://www.absorblearning.com/media/item.action?quick=uh
Movie
http://www.youtube.com/watch?v=w-XbxSiDQWY&feature=PlayList&p=C470A9E10C6186FC&playnext=1&playnext_from=PL&index=45
Another clip about electrolysis
http://www.youtube.com/watch?v=i9xS9t-KMpc&feature=PlayList&p=AA571057652E55EB&index=3
Aluminium extraction simulation
http://www.absorblearning.com/media/attachment.action?quick=u8&att=2166
To learn about this topic
Go to:
http://www.absorblearning.com/
Log in:
Username: student18951
password; science
Go to absorb chemistry
All the content is covered in the sections shown below:
Go to:
http://www.absorblearning.com/
Log in:
Username: student18951
password; science
Go to absorb chemistry
All the content is covered in the sections shown below:
| electrolysis_review.docx | |
| File Size: | 64 kb |
| File Type: | docx |
| topic5electricityandchemistry.docx | |
| File Size: | 6 kb |
| File Type: | docx |

