Topic 7 Chemical reaction
Topic content:
7.1 Rate of reaction
The effect of concentration, particle size, catalysts (including enzymes) and temperature of the rate of reactions
Describe practical methods of measuring the rate of reactions that involve production of gases
Describe suitable methods for investigating the effect of a given variable on the rate of reaction
Interpret data obtained from experiments on reaction rates
Explain the effects of temperature and concentration in terms of collisions between reacting particles.
Apply the factors affecting reaction rate to explosive combustion with fine powders (flour mills) and gases (in mines)
Describe the effect of light in photochemical reactions.
Describe the use of silver salts in photography as the reduction of silver ions to silver and photosynthesis as the reaction between carbon dioxide and water in the presence of chlorophyll and sunlight to produce glucose and oxygen
7.2 Reversible reactions
The idea that some chemical reactions can be reversed by changing the conditions.
The effect of changing the conditions (concentration, temperature and pressure) on reversible reactions.
Concept of equilibrium
7.3 Redox
Define oxidation and reduction in terms of oxygen loss and gain and in terms of electron transfer.
Using oxidation states in naming compounds.
Identify redox reactions by changes in oxidation state and by colour changes involved when using acidified potassium manganate (VII) and potassium iodide
7.1 Rate of reaction
The effect of concentration, particle size, catalysts (including enzymes) and temperature of the rate of reactions
Describe practical methods of measuring the rate of reactions that involve production of gases
Describe suitable methods for investigating the effect of a given variable on the rate of reaction
Interpret data obtained from experiments on reaction rates
Explain the effects of temperature and concentration in terms of collisions between reacting particles.
Apply the factors affecting reaction rate to explosive combustion with fine powders (flour mills) and gases (in mines)
Describe the effect of light in photochemical reactions.
Describe the use of silver salts in photography as the reduction of silver ions to silver and photosynthesis as the reaction between carbon dioxide and water in the presence of chlorophyll and sunlight to produce glucose and oxygen
7.2 Reversible reactions
The idea that some chemical reactions can be reversed by changing the conditions.
The effect of changing the conditions (concentration, temperature and pressure) on reversible reactions.
Concept of equilibrium
7.3 Redox
Define oxidation and reduction in terms of oxygen loss and gain and in terms of electron transfer.
Using oxidation states in naming compounds.
Identify redox reactions by changes in oxidation state and by colour changes involved when using acidified potassium manganate (VII) and potassium iodide
|
|
|
|
Learning activities:
Interactive lesson on reaction rates:http://lgfl.skoool.co.uk/content/keystage4/chemistry/pc/lessons/uk_ks4_rates_of_reaction/h-frame-ns6.htm
Measuring reaction rates of reactions of gases 1:http://www.absorblearning.com/media/attachment.action?quick=vp&att=2272
Measuring reaction rates of reactions of gases 2:http://www.absorblearning.com/media/attachment.action?quick=vo&att=2270
Reaction rates and concentration: http://www.absorblearning.com/media/attachment.action?quick=vx&att=2288
Reaction rates and temperature:http://www.absorblearning.com/media/attachment.action?quick=w2&att=2298
Reaction rates and surface area: http://www.absorblearning.com/media/attachment.action?quick=vw&att=2286
Reaction rates and catalysts: http://www.absorblearning.com/media/attachment.action?quick=w3&att=2300
Explaining reaction rates and catalysts:http://www.absorblearning.com/media/attachment.action?quick=w4&att=2302
Interactive lesson on reaction rates:http://lgfl.skoool.co.uk/content/keystage4/chemistry/pc/lessons/uk_ks4_rates_of_reaction/h-frame-ns6.htm
Measuring reaction rates of reactions of gases 1:http://www.absorblearning.com/media/attachment.action?quick=vp&att=2272
Measuring reaction rates of reactions of gases 2:http://www.absorblearning.com/media/attachment.action?quick=vo&att=2270
Reaction rates and concentration: http://www.absorblearning.com/media/attachment.action?quick=vx&att=2288
Reaction rates and temperature:http://www.absorblearning.com/media/attachment.action?quick=w2&att=2298
Reaction rates and surface area: http://www.absorblearning.com/media/attachment.action?quick=vw&att=2286
Reaction rates and catalysts: http://www.absorblearning.com/media/attachment.action?quick=w3&att=2300
Explaining reaction rates and catalysts:http://www.absorblearning.com/media/attachment.action?quick=w4&att=2302
| topic7chemicalreactions.docx | |
| File Size: | 6 kb |
| File Type: | docx |
Key words
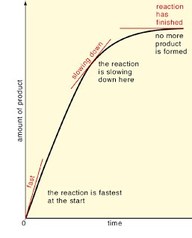
Rate of reaction: is how fast the products in a reaction are made or how quickly the reactants are used up.
Rate = change in concentration of reactant (or product)
time
Catalyst: is a substance that increases the speed of a reaction, without being changed or used up in the reaction.
Surface area: refers to the size of the solid particles in a reaction. The smaller the pieces it is broken into the more surface will be in contact with the solution around it and the bigger surface area it will have (increasing the rate of reaction)
Collision theory: particles have to collide with other and with enough energy in order to react.
Photochemical reaction: a reaction in which the rate can depend upon light intensity
Reversible reaction: a reaction that can go in either direction backwards and forwards
Equilibrium: this is when the rates of the forward reaction and the reverse reaction are equal in a reversible reaction
Oxidation: a chemical reaction in which a substance gains oxygen, loses hydrogen or loses electrons
Reduction: when a substance loses oxygen, gains hydrogen or gain electrons
Redox reaction: any reaction in which electrons are transferred
Rate of reaction: is how fast the products in a reaction are made or how quickly the reactants are used up.
Rate = change in concentration of reactant (or product)
time
Catalyst: is a substance that increases the speed of a reaction, without being changed or used up in the reaction.
Surface area: refers to the size of the solid particles in a reaction. The smaller the pieces it is broken into the more surface will be in contact with the solution around it and the bigger surface area it will have (increasing the rate of reaction)
Collision theory: particles have to collide with other and with enough energy in order to react.
Photochemical reaction: a reaction in which the rate can depend upon light intensity
Reversible reaction: a reaction that can go in either direction backwards and forwards
Equilibrium: this is when the rates of the forward reaction and the reverse reaction are equal in a reversible reaction
Oxidation: a chemical reaction in which a substance gains oxygen, loses hydrogen or loses electrons
Reduction: when a substance loses oxygen, gains hydrogen or gain electrons
Redox reaction: any reaction in which electrons are transferred
| c7_chemical_reactions.pdf | |
| File Size: | 644 kb |
| File Type: | |
| c7_chemical_reactions.doc | |
| File Size: | 952 kb |
| File Type: | doc |