Topic 1: The Particulate Nature of Matter
Topic Content - Core:
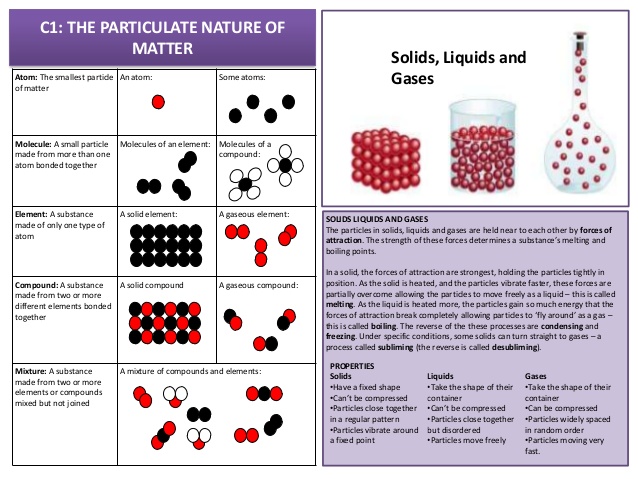
- State the distinguishing properties of solids, liquids and gases
- Describe the structure of solids, liquids and gases in terms of particle separation, arrangement and types of motion
- Describe changes of state in terms of melting, boiling, evaporation, freezing, condensation and sublimation
- Describe qualitatively the pressure and temperature of a gas in terms of the motion of its particles
- Show an understanding of the random motion of particles in a suspension (sometimes known as Brownian motion) as evidence for the kinetic particle (atoms, molecules or ions) model of matter
- Describe and explain diffusion
Topic Content - Supplement
- Explain changes of state in terms of the kinetic theory
- Describe and explain Brownian motion in terms of random molecular bombardment
- State evidence for Brownian motion
- Describe and explain dependence of rate of diffusion on molecular mass
IGCSE Chemistry Key words Topic 1: The particulate nature of matter
- Kinetic particle theory: a theory used to describe the properties of the different states of matter in terms of the movement of particles(atoms or molecules)
- States of matter: solid, liquid and gas-the three states of matter in which any substance can exist, depending on the conditions of temperature and pressure
- Diffusion: the process in which fluids (liquids and gases) mix as a result of the random movement of their particles
Watch these videos to help you to understand:
|
|
|