Topic 13 Carbonates

Topic content:
Describe the manufacture of lime (calcium oxide) from calcium carbonate (limestone) in terms of thermal decomposition.
Name some uses of lime and slaked lime in treating acid soil and neutralizing acidic industrial waste.
Name the uses of calcium carbonate in the manufacture of iron and cement
Describe the manufacture of lime (calcium oxide) from calcium carbonate (limestone) in terms of thermal decomposition.
Name some uses of lime and slaked lime in treating acid soil and neutralizing acidic industrial waste.
Name the uses of calcium carbonate in the manufacture of iron and cement
Learning resources:
Lime Kiln: http://www.absorblearning.com/media/attachment.action?quick=11d&att=2679
Details about lime (calcium oxide):http://www.gcsescience.com/rk20.h
Lime Kiln: http://www.absorblearning.com/media/attachment.action?quick=11d&att=2679
Details about lime (calcium oxide):http://www.gcsescience.com/rk20.h
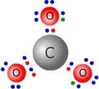
Challenge yourself!
Look at the electron arrangement of the carbonate ion can you work out why this is the structure
Look at the electron arrangement of the carbonate ion can you work out why this is the structure
Key Words: Carbonates
Thermal decomposition: the breaking down of a compound by heating it
Carbonate: compounds which contain the carbonate ion (CO32- )
Carbonate: compounds which contain the carbonate ion (CO32- )
| topic13carbonates.docx | |
| File Size: | 5 kb |
| File Type: | docx |
| c13_carbonates.docx | |
| File Size: | 251 kb |
| File Type: | docx |
| igcse_coursework_on_carbonates.docx | |
| File Size: | 18 kb |
| File Type: | docx |