Topic 2: Experimental techniques
Topic Content 2.1 Measurement - Core
- Name appropriate apparatus for the measurement of time, temperature, mass and volume, including burettes, pipettes and measuring cylinders
Time
Temperature
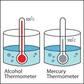
- Measured with thermometer. There 2 types: a) Mercury-in-glass b) Alcohol-in-glass.
- SI Unit: Kelvin (K)
- Daily life measurement: degree Celcius (oC) , K = oC + 273
Mass and Volume
|
Volume is the quantity of three-dimensional space occupied by a liquid, solid, or gas. Common units used to express volume include liters, cubic meters, gallons, milliliters, teaspoons and ounces. Many other units exist.
|
Measuring beakers, pipettes and burettes are used to measure liquids
Topic Content 2.2.1 Criteria of Purity - Core
- Demonstrate knowledge and understanding of paper chromatography
- Interpret simple chromatograms
- Identify substances and assess their purity from melting point and boiling point information
- Understand the importance of purity in substances in everyday life, e.g. foodstuffs and drugs
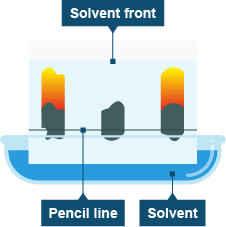
What is Chromatography?Chromatography is a process in which a chemical mixture carried by a liquid or gas is separated into its components. Chromatography can be used to separate mixtures of coloured compounds. Mixtures that are suitable for separation by chromatography include inks, dyes and colouring agents in food.
1. What is Paper Chromatography?Paper Chromatography is a separation technique that is used to separate and identify the components of a mixture. You can learn more about paper chromatography by clicking here.
|
|
2. Interpret Simple Chromatograms
3. Identify Substances and Assess Their Purity From Melting Point and Boiling Point Information
- Pure substances have a fixed boiling and melting point. For example, pure water boils at 100oC and melts at 0oC.
- Impure substances have no fixed melting and boiling points. They can melt and boil at a range of temperatures.
- Also, it can vary the melting and boiling points of pure substances. e.g. pure water boil at 100oC, but when salt is added this increases to 102oC
- The purity of a compound can be established using data from its melting point or boiling point and from thin layer chromatography.
4. Understand the importance of purity in substances in everyday life, e.g. foodstuffs and drugs
Topic Content 2.2.1 Criteria of Purity - Supplement
- Interpret simple chromatograms, including the use of Rf values
- Outline how chromatography techniques can be applied to colourless substances by exposing chromatograms to substances called locating agents (Knowledge of specific locating agents is not required.)
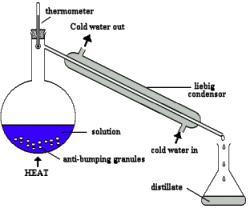
Topic Content 2.2.2 Methods of purification - Core
|
|
Key Words
- Melting point: the temperature at which a solid changes into a liquid
- Boiling point: The temperature at which a liquid turns into a gas
- Volatile: a substance that changes easily into a vapour. It has a low boiling point
- Solution: a mixture of a solute, usually a solid dissolved in the solvent, a liquid
- Locating agent: a substance that reacts with colourless spots on a chromatogram to make them visible as coloured spots
- Rf values (retention factor) : = distance moved by subtance
Try this quick test on separation techniques - click here
Stop! You should now be able to do the following.
Please ask me if there is anything that you don't understand or can't do
- name appropriate apparatus for the measurement of time, temperature, mass and volume, including burettes, pipettes and measuring cylinders
- describe paper chromatography (including the use of locating agents) and interpret simple chromatograms
- indicate that mixtures melt and boil over a range of temperatures
- identify pure substances by distinctive melting and boiling points
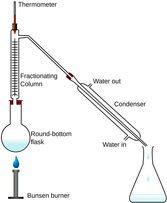
- describe methods of purification by the use of a suitable solvent, filtration, crystallisation, re-crystallisation and distillation (including use of a fractionating column)
- describe petroleum as a mixture of hydrocarbons and its separation into useful fractions by fractional distillation
- name the uses of the fractions: petrol fraction as fuel in cars; paraffin fraction for oil stoves and aircraft fuel; diesel fraction for fuel in diesel engines; lubricating fraction for lubricants; making waxes and polishes and bitumen for making roads.
Key Words
| topic2experimentaltechniques.docx | |
| File Size: | 94 kb |
| File Type: | docx |
| topic2purity.doc | |
| File Size: | 1874 kb |
| File Type: | doc |