Topic 3: Atoms Elements and Compounds
Link to Task Page
Topic content:
3.1 Atomic structure and the periodic table
The relative charges and masses of protons, neutrons and electrons
Define proton number and neutron number
Use proton number and the structure of atoms to explain the basis of the periodic table with particular reference to elements 1 to 20
Define isotopes, state the two types of isotopes radioactive and non-radioactive
State one medical and one industrial use of radioactive isotopes
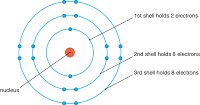
Describe the build up of electrons in shells and understand the significance of the noble gas electronic structure and valency electrons
Topic content:
3.1 Atomic structure and the periodic table
The relative charges and masses of protons, neutrons and electrons
Define proton number and neutron number
Use proton number and the structure of atoms to explain the basis of the periodic table with particular reference to elements 1 to 20
Define isotopes, state the two types of isotopes radioactive and non-radioactive
State one medical and one industrial use of radioactive isotopes
Describe the build up of electrons in shells and understand the significance of the noble gas electronic structure and valency electrons
3.2 Bonding: the structure of matter
The differences between elements, mixtures and compounds and between metals and non-metals
Describe an alloy such as brass as a mixture of a metal with other elements.
3.2 (a) Ions and ionic bonds
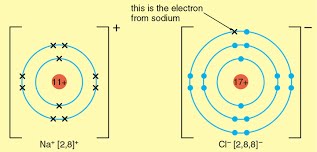
The formation of ions by electron loss or gain.
The formation of ionic bonds between group 1 and group 7 elements and generally between metal and non-metal elements
The lattice structure if ionic compounds as a regular arrangement of alternating positive and negative ions
The formation of ions by electron loss or gain.
The formation of ionic bonds between group 1 and group 7 elements and generally between metal and non-metal elements
The lattice structure if ionic compounds as a regular arrangement of alternating positive and negative ions
3.2 (b)Molecules and covalent bonds
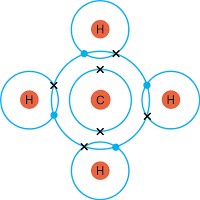
The formation of single covalent bonds in hydrogen, chlorine, water, methane and hydrogen chloride leading to the formation of the noble gas structure.
The electron arrangement in more complex molecules such as nitrogen, ethene, methanol and carbon dioxide.
The differences in volatility, solubility and electrical conductivity between ionic and covalent compounds.
The formation of single covalent bonds in hydrogen, chlorine, water, methane and hydrogen chloride leading to the formation of the noble gas structure.
The electron arrangement in more complex molecules such as nitrogen, ethene, methanol and carbon dioxide.
The differences in volatility, solubility and electrical conductivity between ionic and covalent compounds.
3.2(c) Macromolecules
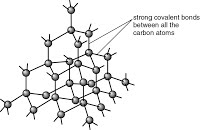
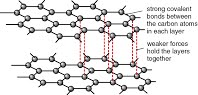
The giant covalent structures of diamond and graphite and how this relates to their respective uses in cutting and as a lubricant
The structure of silicon (VI) oxide and describe the similarity in its properties to the properties of diamon
The giant covalent structures of diamond and graphite and how this relates to their respective uses in cutting and as a lubricant
The structure of silicon (VI) oxide and describe the similarity in its properties to the properties of diamon
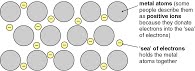
3.2 (d) Metallic bonding
Metallic bonding is a lattice of positive ions in a sea of electrons and how this explains the conductivity and malleability of metals
What are the following showing
Metallic bonding is a lattice of positive ions in a sea of electrons and how this explains the conductivity and malleability of metals
What are the following showing
| topic3atomselementsandcompounds.docx | |
| File Size: | 7 kb |
| File Type: | docx |
Learning resources:
Interactive lesson:http://lgfl.skoool.co.uk/content/keystage4/chemistry/pc/lessons/uk_ks4_structures_and_bonding/h-frame-ns6.htm
Atomic structures of first 20 elements:http://www.absorblearning.com/media/attachment.action?quick=sh&att=2040
Ionic bond forming in sodium chloride:http://www.absorblearning.com/media/attachment.action?quick=sn&att=2052
Covalent bond forming in chlorine: http://www.absorblearning.com/media/attachment.action?quick=sw&att=2070
Covalent bond forming in water: http://www.absorblearning.com/media/attachment.action?quick=su&att=2066
Covalent bond forming in methane:http://www.absorblearning.com/media/attachment.action?quick=sv&att=2068
Covalent bond forming in carbon dioxide:http://www.absorblearning.com/media/attachment.action?quick=sy&att=2074
Giant ionic lattice sodium chloride:http://www.absorblearning.com/media/attachment.action?quick=sp&att=2056
Interactive lesson:http://lgfl.skoool.co.uk/content/keystage4/chemistry/pc/lessons/uk_ks4_structures_and_bonding/h-frame-ns6.htm
Atomic structures of first 20 elements:http://www.absorblearning.com/media/attachment.action?quick=sh&att=2040
Ionic bond forming in sodium chloride:http://www.absorblearning.com/media/attachment.action?quick=sn&att=2052
Covalent bond forming in chlorine: http://www.absorblearning.com/media/attachment.action?quick=sw&att=2070
Covalent bond forming in water: http://www.absorblearning.com/media/attachment.action?quick=su&att=2066
Covalent bond forming in methane:http://www.absorblearning.com/media/attachment.action?quick=sv&att=2068
Covalent bond forming in carbon dioxide:http://www.absorblearning.com/media/attachment.action?quick=sy&att=2074
Giant ionic lattice sodium chloride:http://www.absorblearning.com/media/attachment.action?quick=sp&att=2056
To learn about this topic
Go to:
http://www.absorblearning.com/
Log in:
Username: student18951
password; science
Go to absorb chemistry
All the content is covered in the sections shown below:
Go to:
http://www.absorblearning.com/
Log in:
Username: student18951
password; science
Go to absorb chemistry
All the content is covered in the sections shown below:
Silicon dioxide is not covered in the structures of covalent substances. What you need to know is shown in the image
| questions_practice_year_10.docx | |
| File Size: | 450 kb |
| File Type: | docx |
| c3atomselementsandcompounds.doc | |
| File Size: | 3439 kb |
| File Type: | doc |